Air was drawn slowly and continuously through the solution by connecting the side arm of the test tube to a vacuum source (aspirator or hood vacuum) by way of a water trap using appropriate tubing, Figure 4. The test tube was then clamped in a hot water bath. A Pasteur pipet was inserted through a rubber stopper with a drill hole, to serve as air inlet, Figure 3. Dropwise addition of 0.300 mL of a 30% H 2O 2 solution was then done, followed with dropwise addition of 0.600 mL of concentrated HCl. 6H 2O (300 mg, 1.26 mmol), water (2.5 mL), and 10% ethylenediamine (1 mL) were added to the side arm test tube. Synthesis of T rans-Dichloro bis(ethylenediamine)co-balt(III) Chloride A modified procedure from Szafran, Pike, and Singh for the synthesis of trans-dichloro bis(ethylenediamine) cobalt (III) chloride, trans-Cl, was performed. Co 2+ + H + + H 2O 2 → Co 3+ + H 2O Which species is the oxidizing agent? Which is the reducing agent?įig ure 2. 2) Balance the oxidation-reduction reaction involved in the synthesis of trans-Cl. Students were provided with an experimental handout and asked to answer the following prelab questions: 1) Draw the structure of the complexes, trans-Cl and cis-Cl 2. The cuvette is immersed into hot water bath and transferred every 2 minutes to the UV-Vis spectrophotometer for a wavelength scan. Thermal monitoring of the ligand substitution was carried out using Cary-60 UV-Vis spectrophotometer. Reagents were obtained from commercial sources and used as received. Alternatively, these can be purchased from several manufacturers such as Eisco labs, Haines Educational, and OnlineScienceMall, among others. Experimental Section Customized side arm test tubes ( Figure 2) were fabricated at Illinois College Department of Chemistry by the laboratory manager, Andy Stice. Upper level undergraduate students can be asked to calculate the rate constant for the ligand exchange reaction of water for a chloride ion in trans-dichlorobis(ethylenediamine)cobalt(III) ion, with the assumption of a pseudo-first order rate law. The synthesis of trans complex will be done during the first meeting, then characterization of trans-Cl and aquation will be on the second week. This is a two-week laboratory experiment. 
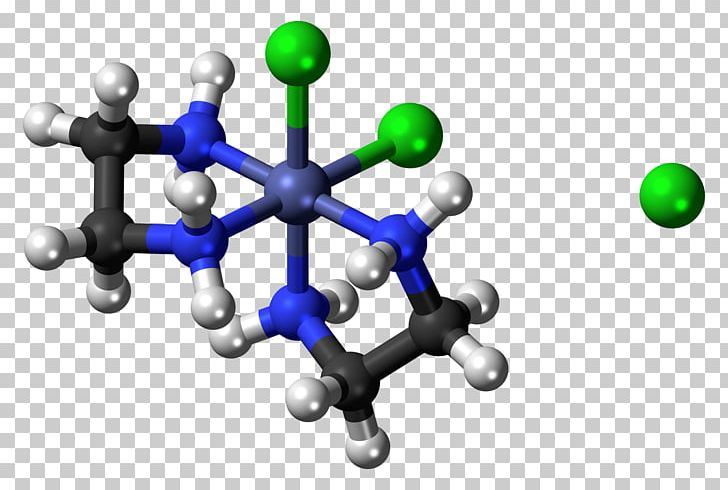
It also provides students the opportunity to see the energy and stability difference of geometric isomers. This laboratory experiment introduces inorganic chemistry undergraduate students to the synthesis and characterization of coordination compounds, geometric isomerism, ligand substitution, magnetism and d n electron configuration, and the use of UV-Vis spectroscopy for monitoring reaction progress. Example of geometric isomers with octahedral geometry The aquation product, Cl 2, is purple in color and has cis geometry. This allows us to monitor the substitution of chloride by water in the green trans-dichloro bis(ethylenediamine)cobalt(III) chloride, trans-Cl complex. Due to the relative inertness of octahedral cobalt(III) metal centers, their ligand substitution and isomerization reactions are slow compared to the complexes of many other transition metals. Octahedral cobalt(III) complexes that are low-spin d 6 configuration are diamagnetic and considered stable, having a filled t 2g subshell. Figure 1 shows an example of octahedral geometric isomers. Introduction Disubstituted planar and octahedral complexes exhibit both cis and trans stereochemistry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
